MEDICAL DEVICE AUTHORITY (MDA) PARTICIPATES IN WHO COLLABORATIVE REGISTRATION PROCEDURE (CRP) FOR PREQUALIFIED IN VITRO DIAGNOSTICS

ANNOUNCEMENT UPDATE
Medical Device Authority (MDA) Participates in WHO Collaborative Registration Procedure (CRP) for Prequalified In Vitro Diagnostics
Dear All Medical Device Industry Stakeholders,
The Medical Device Authority (MDA), Ministry of Health Malaysia is pleased to announce that effective 3 February 2026, MDA has officially become a participating National Regulatory Authority (NRA) in the World Health Organization Collaborative Registration Procedure (CRP) for WHO-prequalified in vitro diagnostics (IVDs).
This reliance-based regulatory mechanism is designed to facilitate and accelerate national registration by leveraging WHO prequalification assessments. It represents a significant step in reducing duplicative regulatory work and ensuring timely access to quality-assured diagnostics.
Manufacturers or applicants who intend to utilize this pathway are required to comply with the national regulatory requirements and submission procedures established by MDA, in addition to the relevant requirements of the WHO CRP.
Eligibility Criteria
To qualify for the CRP pathway in Malaysia, applicants must adhere to the following criteria:
|
Requirement |
Details |
|
Scope |
· Manufacturers or authorized representatives seeking registration of WHO-prequalified IVDs in Malaysia. · IVD products that have been successfully prequalified by WHO. · Applications where the applicant intends to use the Collaborative Registration Procedure (CRP) to facilitate national registration. |
|
WHO Prequalification Status |
The IVD product must be successfully prequalified and listed under the WHO Prequalification Programme. |
|
Product Sameness |
The submitted product must be identical in all essential aspects to the WHO-prequalified version. |
|
Manufacturer Consent |
The manufacturer must provide written consent to WHO to share relevant confidential assessment reports with MDA. |
|
Compliance with Malaysian Regulations |
The applicant must comply with all applicable Malaysian regulatory requirements, submission procedures, and fee payments. |
Implementation Pathway
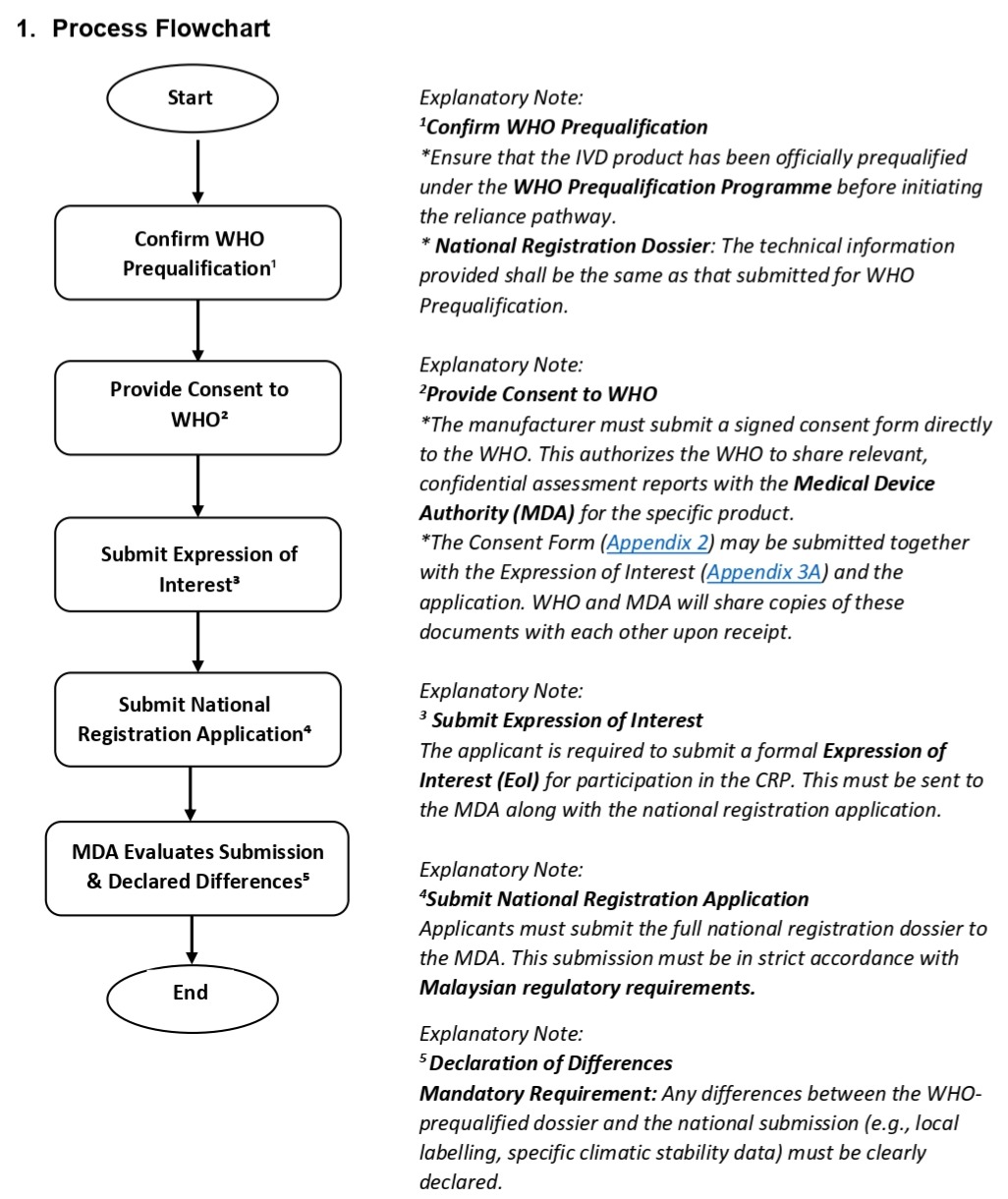
Manufacturers of WHO-prequalified in vitro diagnostic medical devices who wish to apply for registration in Malaysia using the CRP should follow the steps below:
Review Timeline
Under the Collaborative Registration Procedure, participating NRAs aim to complete the regulatory review within 90 working days after receiving access to all relevant information from WHO. The application review process will begin once a complete application has been submitted and all relevant information from WHO has been received.
Applicants should note that the timeline may vary depending on:
- Completeness of the submission
- Any additional information requested by MDA
- National administrative processes
Important Notes
- Participation in the CRP is voluntary.
- National regulatory requirements and applicable fees remain applicable.
- MDA retains full authority to make the final regulatory decision regarding product registration in Malaysia.
- Any post-registration changes to the registered product must be reported through the CRP. All such changes should be submitted to both WHO and MDA in accordance with Section 6 of the CRP IVDs Procedure.
Further Information
For additional information regarding the Collaborative Registration Procedure, please refer to the World Health Organization website.
- TRs 1060 - Annex 7: Good practices of national regulatory authorities in implementing the collaborative registration procedures for medical products
- https://www.who.int/publications/m/item/collaborative-procedure-between-the-who-and-nra-s-in-the-assessment-and-accelerated-national-registration-of-who-prequalified-ivd-s-annex4
For enquiries related to CRP submissions in Malaysia, applicants may contact:
Medical Device Authority (MDA)
Ministry of Health Malaysia
Website: https://www.mda.gov.my
Email: [email protected]