Appendix 3: Technology Readiness Level Chart

Appendix 2: Innovative Medical Device Designation Criteria
Innovative Medical Device Designation Criteria
This section outlines the eligibility for a medical device to be classified as an innovative medical device according to the following criteria:
- The device should demonstrate novel technology or a significant improvement in design, effectiveness, or efficiency compared to existing devices, including but not limited to the application of AI, additive manufacturing (e.g., 3D printing), or other advanced technologies, and shall be supported, where applicable, by a valid patent or documented intellectual property;
- The proposed device should fall within Technology Readiness Level (TRL) 3 to 9, indicating progression from proof-of-concept to system validation and readiness for deployment; and
- The innovative medical device should have a well-established proof of concept.
Note 1: TRL 3 refers to Proof of Concept (POC), where the device has undergone basic testing to verify that the concept works in a controlled environment.
Note 2: TRL 4 to TRL 6 involve the development and testing of prototypes, while TRL 7 to TRL 9 represent stages where the device is ready for market use or is already operational in the field.
Appendix 1: Definition Of A Medical Device As Outlined Under Section 2 Of Act 737
Definition Of A Medical Device As Outlined Under Section 2 Of Act 737
Section 2 defines a “medical device” as any instrument, apparatus, implement, machine, appliance, implant, in vitro reagent or calibrator, software, material, or other similar or related article intended by the manufacturer to be used, alone or in combination, for human beings for the purpose of:
- diagnosis, prevention, monitoring, treatment, or alleviation of disease;
- diagnosis, monitoring, treatment, alleviation of, or compensation for an injury;
- investigation, replacement, modification, or support of the anatomy or a physiological process;
- supporting or sustaining life;
- control of conception;
- disinfection of a medical device; or
- providing information for medical or diagnostic purposes by means of in vitro examination of specimens derived from the human body
INNOVATIVE MEDICAL DEVICE REVIEW (IMDR) APPLICATION
INNOVATIVE MEDICAL DEVICE REVIEW (IMDR) APPLICATION
INTRODUCTION
An innovative medical device is commonly defined as a device that introduces a novel idea in technology use or performance. It incorporates new technology or a significant improvement in design, effectiveness, or efficacy compared to existing medical devices, demonstrating meaningful clinical benefits and addressing identified unmet clinical needs.
To facilitate the growth of innovative medical devices, the Medical Device Authority has established a step-by-step application process for Innovative Medical Device Review (IMDR) for both industry and academia. As this initiative takes place prior to the placement of a medical device on the market, it is designed to assist relevant parties in understanding expectations for compliance within the regulatory requirements landscape.
Interested parties may refer to the following step-by-step application process and the following reference documents. Application for Innovative Medical Device Review can be made through the application form
Reference Documents
- Innovative Medical Device Review Application Form (Word/ PDF)
- Step-By-Step Process For Innovative Medical Device Review (IMDR) Application
- Appendix 1: Definition Of A Medical Device As Outlined Under Section 2 Of Act 737
- Appendix 2: Innovative Medical Device Designation Criteria
- Appendix 3: Technology Readiness Level Chart
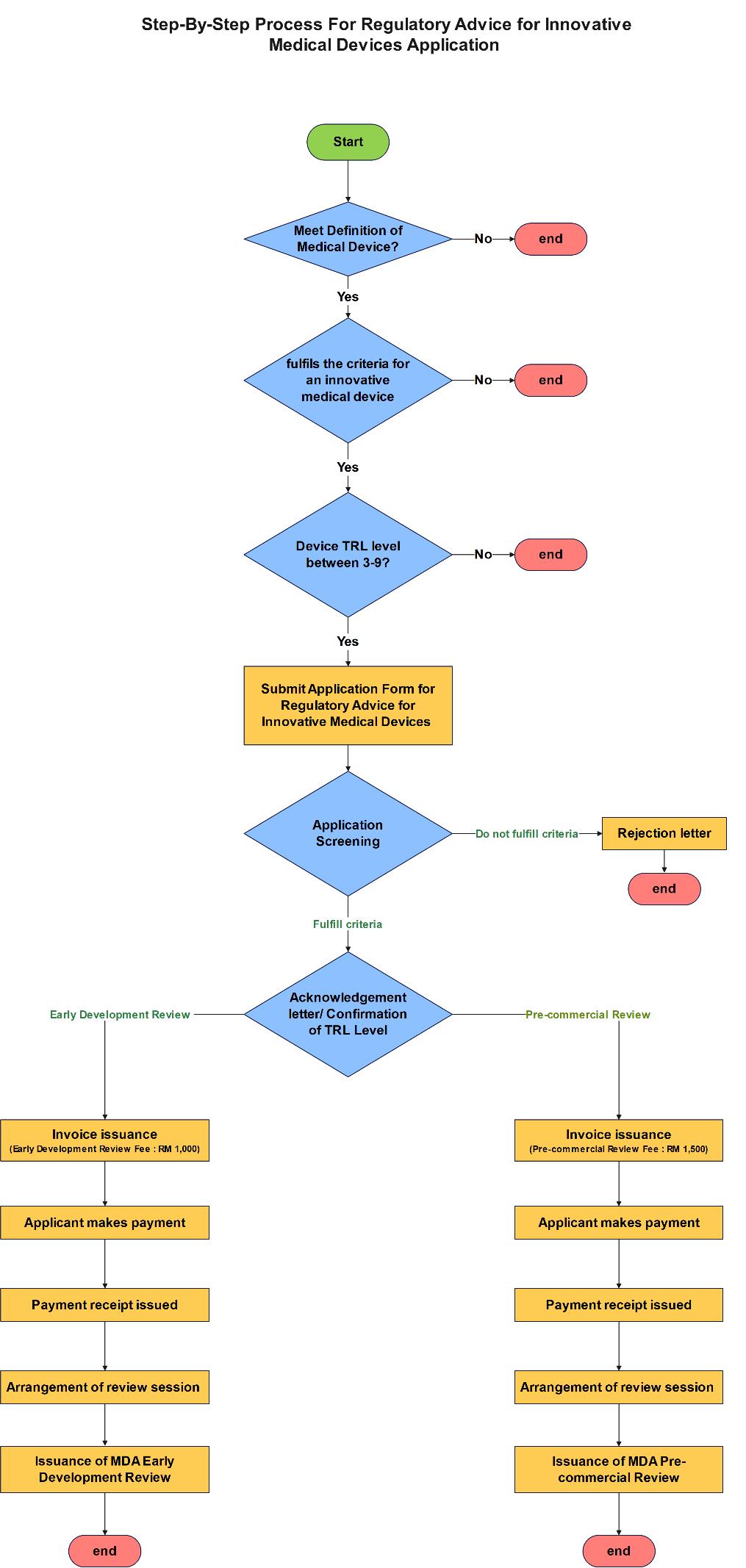
Step-By-Step Process For Innovative Medical Device Review (IMDR) Application
Step-By-Step Process For Innovative Medical Device Review (IMDR) Application
|
Step |
Responsible Party |
Action / Instruction |
Output |
|
1 |
Applicant |
The applicant shall determine whether the product fulfils ALL of the following criteria: a. falls within the definition of a medical device under Section 2, Act 737 b. fulfils the criteria for an innovative medical device c. the device is currently within technology readiness level 3 to 9 as outlined by MDA: i. TRL 3 – TRL 5 (eligible for Early Development Review) ii. TRL 6 – TRL 9 (eligible for Pre-commercial Review) Note 1: Please refer the appendices of this document for the following: i. Appendix 1: Definition Of A Medical Device As Outlined Under Section 2 Of Act 737 ii. Appendix 2: Innovative Medical Device Designation Criteria iii. Technology readiness level chart Note 2: Parties applying for a Pre-Commercial Review must possess a working prototype |
Meet pre-defined criteria |
|
2 |
Applicant |
If the product meets the definition and innovation criteria, the applicant shall complete the Application Form for Regulatory Advice for Innovative Medical Devices and submit the completed form together with all required supporting documents via email to [email protected] . |
Submit Application Form for IMDR |
|
3 |
MDA (Technical Team) |
The MDA Technical Team shall screen the application and determine, based on the information provided, whether the application fulfils the innovation criteria and appropriate TRL level. If the criteria are met, the MDA Technical Team shall issue an Acknowledgement Letter along with the confirmation of the corresponding TRL level. If the criteria are not met, the MDA Technical Team shall issue an Application Rejection Letter. |
Application screening Acknowledgement letter/ Rejection letter |
|
4 |
MDA (Finance Division) |
The MDA Finance Division shall generate an invoice detailing the prescribed application fee and shall email the invoice directly to the applicant. Note: · A processing fee of RM1,000 is prescribed for early development review, applicable to Technology Readiness Levels (TRL) 3–5 · A processing fee of RM1,500 is prescribed for pre-commercialization review, applicable to Technology Readiness Levels (TRL) 6–9 |
Invoice issuance |
|
5A |
Applicant |
The applicant shall make the online payment within 30 days from the date of invoice issuance in accordance with the instructions provided by the MDA Finance Division. Failure by the applicant to make payment within the stipulated time frame will result in the application being dismissed. The applicant will then be required to restart the application process from the beginning |
Applicant makes payment |
|
MDA (Finance Division) |
The MDA Finance Division issues payment receipt |
Payment receipt issued |
|
|
5B |
MDA (Technical Team) / Applicant |
Upon issuance of payment receipt, the MDA Technical Team shall arrange an advisory session for the innovative medical device within 14 working days* and may require the applicant to present the device. The advisory session shall not exceed two (2) hours. Afterwards, MDA Technical Team shall evaluate the application within 30 working days in accordance with the client charter. Note: The advisory session may be rescheduled in the event that either the applicant or the MDA is unavailable. |
Arrangement of review session |
|
6 |
MDA (Technical Team) |
The MDA Technical Team shall issue the MDA Early Development Review (for TRL 3-9) or Pre-Commercialization Review (for TRL 6-9) report based on the evaluation findings and advisory session outcomes. |
Issuance of MDA Early Development Review/ Pre-Commercialization Review Report |
Flow Chart